SHANGHAI, July 22, 2024 /PRNewswire/ — The molecular design strategy and experimental results of Bright Gene’s dual GLP-1/GIP receptor agonist, BGM0504, have been published online in Scientific Reports, a sub-journal of Nature, on July 19, 2024. Bright Gene (Stock Code: 688166.SH) is an innovative pharmaceutical company emerging on the international stage, is focused on developing best-in-class pharmaceuticals to improve patient health globally.
The article, titled “Molecular Dynamics Guided Optimization of BGM0504 Enhances Dual Target Agonism for Combating Diabetes and Obesity”, presents the findings of BGM0504’s development.
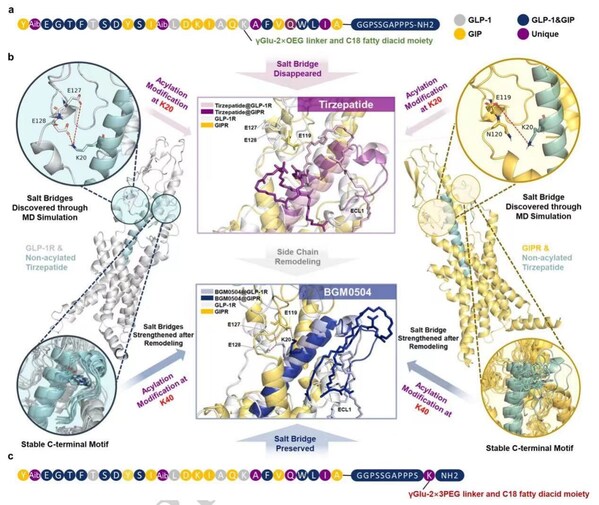
BGM0504, an AI-assisted designed dual GIP and GLP-1 receptor agonist, demonstrates superior efficacy in both in vitro and in vivo experiments. Using AI-driven computer simulations, Bright Gene has discovered that optimal interaction between the glutamate residues on both GLP-1R and GIPR and the K20 residue of a peptide agonist provide superior activity. This interaction is a key insight not evident in cryo-EM studies. BGM0504 was designed to preserve the free amino group of the K20 residue by shifting the acylation point to position 40 of BGM0504. This design resulted in a 3-fold increase in agonistic effects on GLP-1R and GIPR, with superior therapeutic outcomes in diabetic and obesity mouse models.
About Bright Gene and BGM0504
Bright Gene (Stock Code: 688166.SH) is an innovative pharmaceutical company focused on developing best-in-class pharmaceuticals. The company integrates APIs and formulations, combining generic and innovative drugs to meet global clinical needs. BGM0504 is a dual GIP/GLP-1 receptor agonist for treating type 2 diabetes, obesity, and NASH, currently in the late stages of Phase II clinical trials.
Reference
Yuan, J., Liu, W., Jiang, X. et al. Molecular dynamics-guided optimization of BGM0504 enhances dual-target agonism for combating diabetes and obesity. Sci Rep 14, 16680 (2024). https://doi.org/10.1038/s41598-024-66998-8


Đề xuất
Kyowa Kirin and Kura Oncology Initiate Japanese Phase 2 Registration-Directed Trial of Ziftomenib in R/R NPM1-m AML
Media Advisory-Ontario Health Coalition/Large cross-province protests this Saturday: Ford gov’t pushing our hospitals into deficit & cuts while paying more to privatize their services
Changan Group Unveils Updated “Vast Ocean Plan 2.0” and highlight Next-Generation BlueCore Hybrid at Auto China 2026
Kerry Properties Presents Asia’s Signature Ice Hockey Event
HanchorBio Selects Bloomberg to Strengthen its Strategic Intelligence Across Drug Development and Global Biopharma
Dingdong Files Its Annual Report on Form 20-F